views

For over 30 years, we've been doing in vivo pyrogen testing with speed and accuracy, providing quick sample turnaround and technical competence. We use our own specific-pathogen-free rabbits in our specialised Good Manufacturing Practices compliant facilities to undertake this crucial component of our clients' quality control programme. Protein-based, non-protein, and blood-based materials are among the test items. Both IMPs and approved medicinal products can be tested for pyrogenicity.
When testing for non-endotoxin pyrogens and other products for which LAL is insufficient, the rabbit pyrogen test (RPT) remains a suitable mammalian test paradigm. Regulatory authorities may also need one-time pyrogen testing to support endotoxin testing routinely. All in vivo rabbit pyrogen assays follow the USP, European, and/or Japanese Pharmacopeia.
Read More: https://bloggerthrive.blogspot.com/2022/02/what-is-pyrogen-and-pyrogen-testing.html
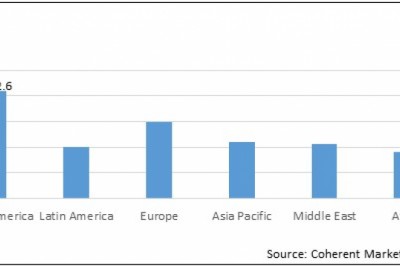
Click here for Pyrogen Testing Market Report:
https://www.coherentmarketinsights.com/market-insight/pyrogen-testing-market-243
Click here for Pyrogen Testing Market Press Release: